 Our rating scale is numerical: one, two, three, four, and five, with five being the highest on the scale.This obscures the affective and playful dimensions that. It features the causes and consequences of a cultural phenomenon that is generally regarded a societal problem and hence focuses (too) much on its dangers (Harambam 2020b, 216). Future research: exploring the affective and playful dimensions of conspiracy theories Research on conspiracy theories is mostly rather serious business.(The Routledge Companion to Media Disinformation and Populism) In each of these conflicts, the overwhelming advantage in firepower. The conflicts in Somalia (1993), Kosovo (1999), Afghanistan (2001-present) and Iraq (2003-201 1) laid bare the limits of conventional military force. ‘The fifth dimension of war’ In the 30 years since the first Gulf War, the nature of war has radically transformed.(Social Computing, Behavioral-Cultural Modeling and Prediction) For example, in some environments a phone may see the same WiFi hotspot repeatedly through the day, while other environments may have an. Entropy Based Footprint Features While the simple frequency features above give some insight into the environment of the phone, they ignore important evidence like the distribution of this activity.This holds for all but the lowest achievable temperatures. A generalisation is : For a macroscopic system with thermal inertia (heat capacity) C » k, the tempergy т is the energy needed to increase the dimensionless entropy by unity. Key Point 6.12 The energy т is required to increase a system’s dimensionless entropy by one at constant temperature. (6.11) and (6.12) that for an isothermal heat process at constant volume, ASdim = Q/r, so if ASdim = 1, then Q = r. Thus, for this simple model of two-state independent particles, r is not a good indicator of the average energy per particle.ĭimensionless entropy and r. Figure 6.7 shows that E/N < 0.28r for all values of r. 
Thus, N ex/N -> 0.5, E/N = N exe/N ->■ 0.5c, and the energy per particle (E/N)/t -> 0 for r -> oo. 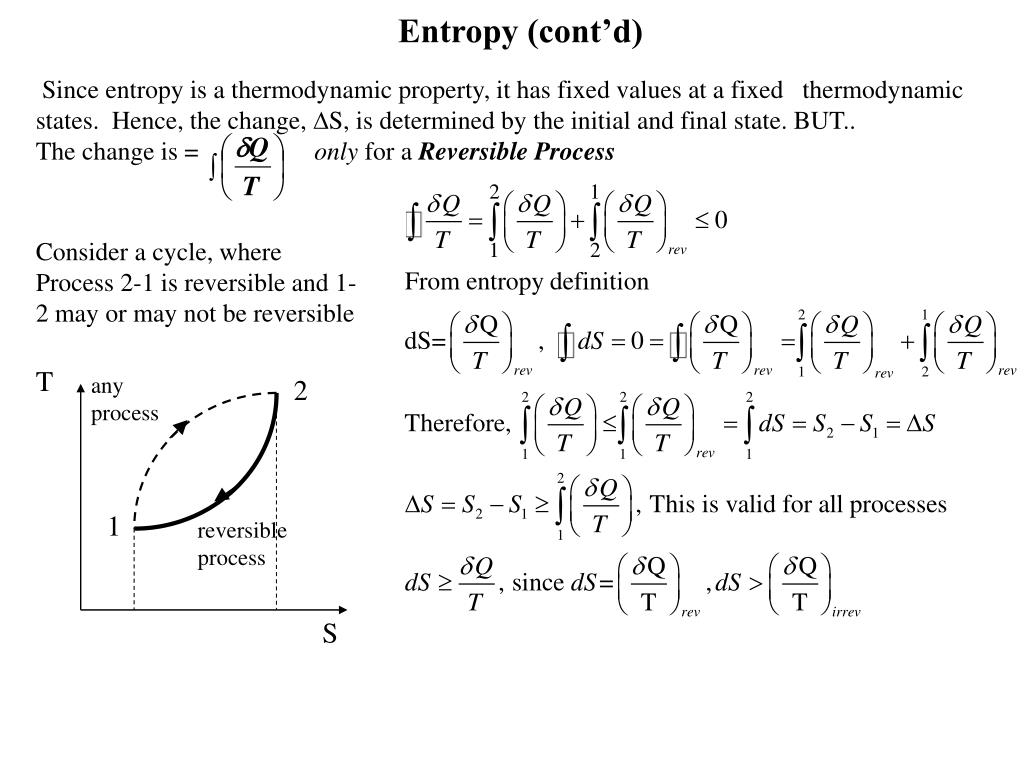
That is, the number of excited st ates with energy e approaches 1/2. For high temperatures, i.e., with т/е > 1, the fraction of particles with energy e approaches 1/2. Next, consider a system with N independent, two-state particles, each with possible energies 0 and e > 0. Tempergy itself is not a good measure of the internal energy per particle for an ideal gas of bosons below the critical temperature. Thus (E/N)/t is not constant, but rather is proportional to r 3/. (4.6) and (4.7), the energy per particle is A helpful result from statistical mechanics is that below the critical temperature, the internal energy per excited particle of an ideal Bose-Einstein gas is E/N ex = 0.77r. It turns out that the dimensionless entropies per particle of monatomic solids are typically A 3 with A 3 = (h 2 /(2тт'ткТ )) 3/ 2. which is consistent with о approaching zero for T (and t) -> 0. At T = 1 K, solid silver has о = 8.5 x 10“ ’. Similarly, one finds that diamond has a = 57.79. (6.13) implies the dimensionless entropy per particle, er = 50.68. What are typical values of o, the dimensionless entropy per molecule? Consider graphite, with molar entropy S mo = 5.7.1 K _1 mol -1 at standard temperature and pressure. Numericsĭimensionless entropy per molecule. Key Point 6.9 If temperature had been defined historically as an energy, entropy would have been dimensionless by definition and we might never have encountered the Kelvin temperature scale or Boltzmann’s constant. Tempergy is intensive, has energy units, and is not related to a stored system energy in general, as I discuss below. 
Internal energy is extensive and represents a stored energy. However they are very different entities physically. The kelvin can be viewed as an energy, i.e., 1 K=l.3807310223 J, and tempergy and internal energy have the same units. The universal gas constant R = 8.3145 J mol 1. Going one step further, define o, the dimensionless entropy per particle, The dimensionless entropy satisfies the property, I prefer to work with the number of molecules N rather than the number of moles. 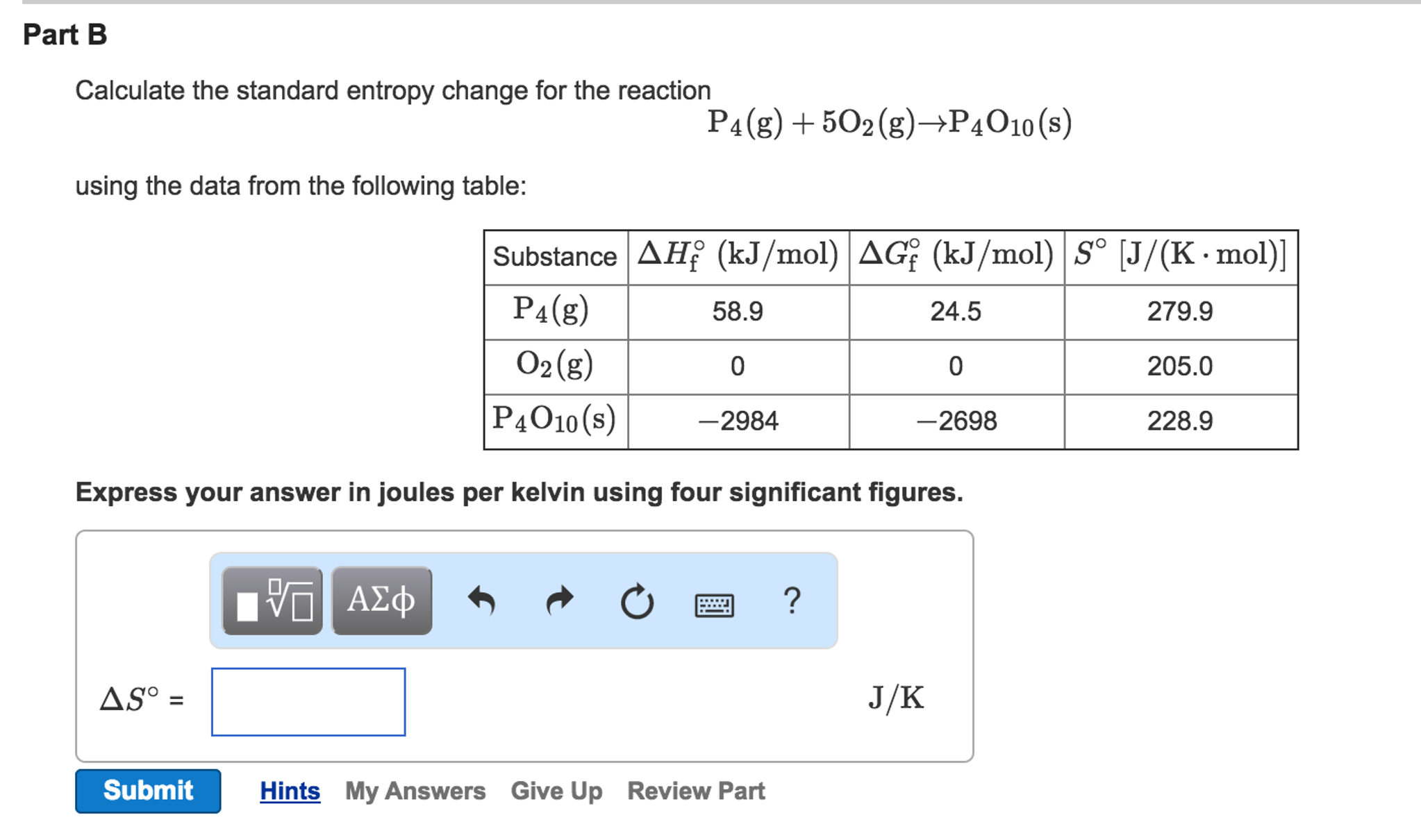
П = N/N a, and N a is Avogadro’s number, is tabulated. Instead, the entropy per mole, 5 moi = S/n, where The entropy 5 is proportional to the system’s number of particles N so it cannot be tabulated numerically in handbooks or databases. (3.3), S(E) = к In Cl(E), the system has internal energy E consistent with its temperature and pressure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
